Findings
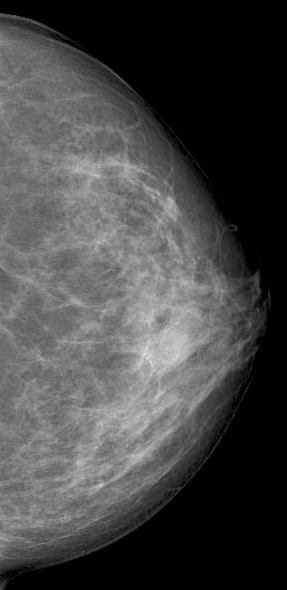
Diagnostic mammography showed scattered fibroglandular densities
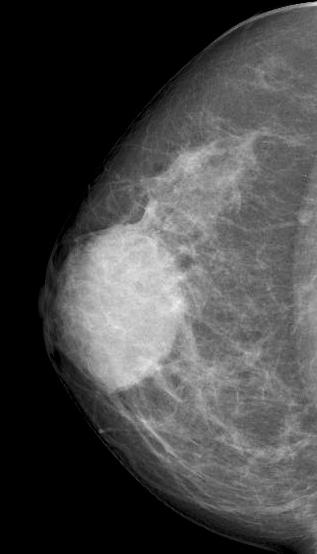
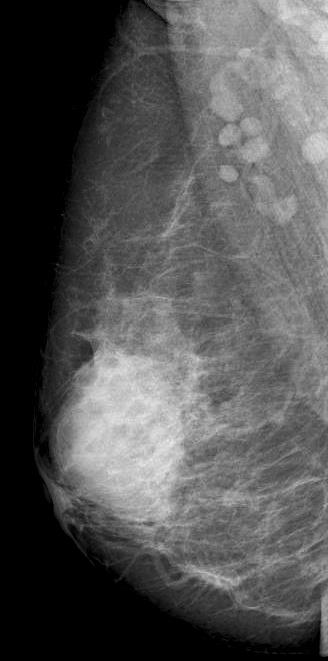
Right breast showed in retroareolar region, an equal density oval mass with partially circumscribed margins measuring 6.6x5.2 cm. There were no associated suspicious calcifications. Right axilla showed a round dense LN of 1 cm.
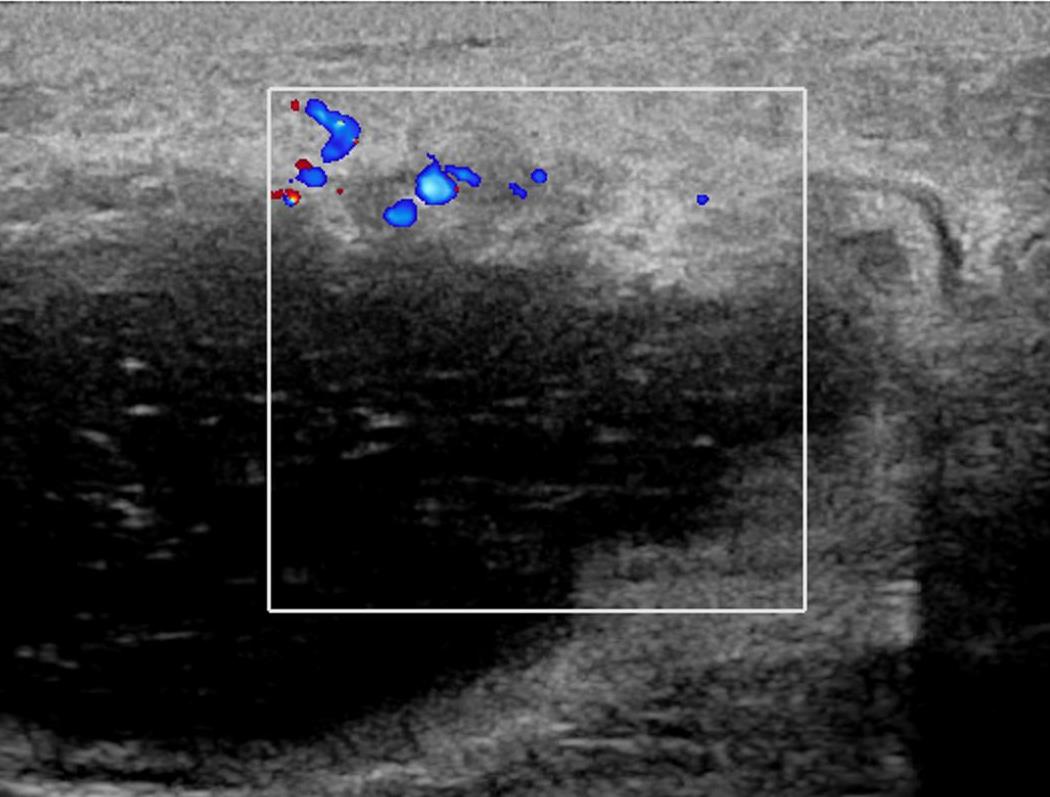
Ultrasound showed at 12 o’clock position in retroareolar region, a complicated cyst with thick nodular wall measuring 4.4x3.1x5.0 cm. Right axilla showed two round LN with effaced hila measuring 0.6 cm.
Core biopsy was performed that showed granulomatous mastitis with focal suppuration. Special stains were negative for AFB and fungi. Fine needle aspiration cytology of the axillary lymph nodes also showed reactive cytology.
After initial response on steroids and aspiration twice, clinical progression of the lesion was noted. Excision biopsy was performed and final pathology confirmed lobulocentric granulomatous inflammation.
Answer
Idiopathic granulomatous mastitis.
Discussion
Idiopathic granulomatous mastitis (IGM) is also known as non-puerperal mastitis or granulomatous lobular mastitis. In 1972, Kessler and Wolloch first described this rare benign chronic inflammatory disease of the breast that is characterised sterile noncaseating lobulocentric granulomatous inflammation.
Pathophysiology: The most common theory postulates initial insult to ductal epithelial cells leads to transition of secretions into the lobular breast stroma. A local inflammatory response is incited in the connective tissue leading to migration of macrophage and lymphocytes into this region with subsequent granuloma formation. Multiple inciting factors contributing to the initial insult have been grouped under mammary duct–associated inflammatory disease sequence (MDAIDS). These include periductal mastitis, mammary duct ectasia, pregnancy, lactation, hyperprolactinemia, α1-antitrypsin deficiency, oral contraceptive use, trauma, diabetes, autoimmune disease, infection with Corynebacterium kroppenstedtii and smoking. However the clinical association has been proved only for pregnancy, lactation and hyperprolactinemia.
Demography: This commonly affects parous women within 5 years of last childbirth with the range from 2 months to 20 years after pregnancy. However, this is also seen in nulliparous females with hyperprolactinemia and rarely in males where the predisposing factors include trauma, smoking and autoimmune diseases.
Clinical manifestation: Wide range of presentations with the most common being tender unilateral breast mass of size varying from 1-20 cm. Isolated skin induration or tender mass with skin erythema and edema are also seen in one third of the cases. In about 20% of the cases, there is unilateral breast enlargement with heaviness that simulates IBC.
Nipple is rarely involved.
Axillary lymphadenopathy is seen in 28% of the patients. Breast neoplasm with possible axillary nodal metastases has to be excluded.
In later stages, abscess formation with or without draining sinuses can form.
Imaging: Depending on patients’ age, clinical manifestations and risk factors, targeted ultrasound, mammography and less commonly MRI breast is performed.
Mammographic features include focal or global asymmetry, irregular focal mass, skin thickening with edema or trabecular thickening, asymmetric increased breast density and axillary lymphadenopathy. It can rarely present as a circumscribed mass or architectural distortion. Calcifications are very rare. Mammogram will be normal in about 8-45% of the cases in the setting of heterogeneously and extremely dense fibroglandular tissue.
Ultrasound commonly shows irregular hypoechoic confluent mass with tubular extensions, circumscribed hypoechoic mass, skin thickening and edema with axillary lymphadenopathy having reactive features. Less common features include heterogeneous hypoechoic confluent masses with indistinct, lobulated or angular margins, parenchymal distortion with no discrete masses and heterogeneous parenchyma with edema. The lesions and surroundings show increased vascularity. The findings will be normal in 4-20% of the cases. With advanced disease, fluid collections or abscesses may be present.
Multiparametric MRI breast findings include T2 hyper intensity of the breast stroma suggesting edema, rim enhancing lesions, heterogeneously enhancing masses with or without non mass enhancement. NME can present in segmental or regional distribution. Kinetic properties are variable. In 20% of the cases it presents as a T2 hyperintense enhancing mass with irregular margins.
Differentials: Idiopathic granulomatous mastitis is basically a disease of exclusion. The differentials include inflammatory breast cancer (IBC), infective mastitis, foreign body injection granulomas, mammary duct ectasia, diabetic fibrous mastopathy and systemic granulomatous processes.
Intervention: Core-needle biopsy, with or without fine-needle aspiration for cytopathology examination, and culture analysis are usually required to exclude IBC and other benign inflammatory breast processes. The sensitivity rate for detection of IGM on core biopsy is 96%. Presence of lobulocentric noncaseating granulomas in combination with a localized infiltration of multi-nucleated giant cells, epithelioid histiocytes, and plasma cells. with negative microbial staining and culture results suggests the possibility of IGM.
Management and Treatment Algorithm: There is an excellent prognosis when diagnosed and treated early. Presence of an abscess at clinical examination and or imaging should not preclude tissue biopsy if the clinical course suggests possible malignancy.
When infectious mastitis is suspected, the initial treatment is with antibiotics. In case of no response, radiological assessment is performed with guided core biopsy and cultures. If there is no IGM, underlying cause is treated.
When IGM is proved with mild symptoms, the patients are offered surveillance. In case of moderate to severe symptoms, corticosteroids are given with tapering dose. With partial or complete response, the patients can be monitored.
If there is contraindication or no response to steroids, methotrexate or bromocriptine can be given. Complete response if achieved can be followed up with routine screening. In case of partial or no response surgery is to be offered.
Breast Imager’s role in IGM:
1. Establish multiplicity and location of the lesions.
2. Document the size of the lesion.
3. Identify abscess formation with possible intervention.
4. Evaluate stability or interval change of the lesions.
5. Evaluate treatment response.
6. Identify metachronous disease and recurrence during treatment.
Reference
1. Kessler E, Wolloch Y: Granulomatous mastitis: a lesion clinically simulating carcinoma. AM J Clin Pathol, 1972; 58: 642–646.
2. Pluguez-Turull CW, Nanyes JE, Quintero CJ, Alizai H, Mais DD, Kist KA, Dornbluth NC. Idiopathic granulomatous mastitis: manifestations at multimodality imaging and pitfalls. Radiographics. 2018 Mar;38(2):330-56.
3. Wolfrum A, Kümmel S, Theuerkauf I, Pelz E, Reinisch M. Granulomatous mastitis: a therapeutic and diagnostic challenge. Breast care. 2018;13(6):413-8.